Food Safety Management Software
High-quality food safety management software that seamlessly integrates with your existing systems to provide a simple, automated and holistic platform for HACCP and FSSC 22000.
• No card required
In an era of rapid and relentless digital transformation of all spheres, the availability of information dominates every business organization.
Our unwavering commitment to flexibility and scalability guarantees smooth integration with existing frameworks while laying the foundation for future expansion and growth.
Try Now
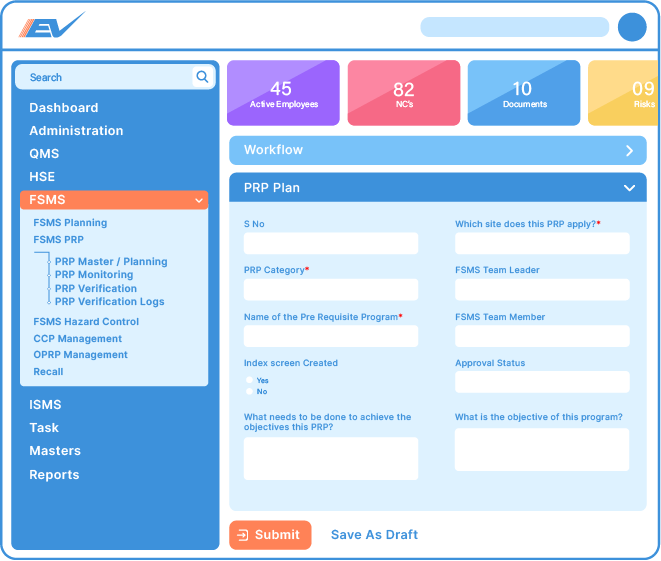
Available in either cloud-based or on-premises formats, Effivity is an off-the-shelf solution for food safety management that can be configured in just 10 minutes even with no prior technical knowledge.
Simple drag-and-drop technology makes this a no-code system that can be installed and updated by anybody.
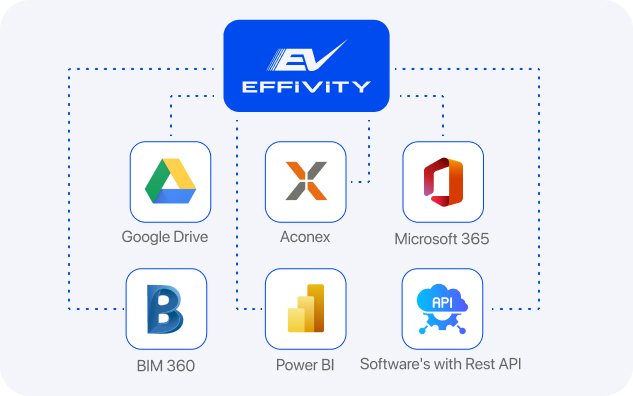
Once integrated with your existing systems Effivity provides real-time actionable insights, automates compliance and enables authorised personnel to view high-level or specific data concerning food safety within your organisation at any time.
• No card required
The food industry is strictly regulated and it’s essential that all businesses involved in the production, transportation, storage or sale of foodstuffs remain fully compliant at all times.
Businesses that fail in their food safety obligations or can’t provide evidence of compliance face the prospect of heavy fines, reputational damage and loss of profits.
Paper-based safety management systems are simply not up to the job of managing modern food safety. They are inaccessible, liable to damage, destruction or input errors, and make internal and external audit procedures laborious and time-consuming
Safety management software simplifies the system, stores all the necessary data in one accessible place and automates reminders. It makes communication simpler and allows for relevant information to be shared quickly and efficiently resulting in far quicker collaboration even in distributed teams.
Food Safety Management System principles run through every aspect of the food industry and must be at the centre of communication, system management, prerequisite programs (PRPs) and Hazard Analysis Critical Control Point (HACCP).
This presents a challenge for business leaders who are trying to keep on top of all these disparate parts of their business. Traditionally, this process has been incredibly time-consuming, labour intensive or overly expensive – sometimes all three.
Effivity food safety management software makes managing multiple aspects of your business simple. It ties together all aspects of food safety, acting as a central repository, automating compliance procedures and giving leaders a bird’s eye view of the organisation
It puts you firmly in control, standardising processes, ensuring consistency in output and productivity, and helping you achieve and maintain food safety certification.
Effivity is a robust, globally proven and effective food safety management software that can be used to meet the required standards for:
✓ ISO 22000:2018
✓ HACCP
✓ BRC
✓ FSSC 22000
✓ BV
✓ BSI
✓ TUV
✓ LRQA
✓ UKAS
✓ ANAB
✓ DAR
✓ JAS-ANZ
✓ NABCB
✓ DAC
✓ SGS
✓ DNV
Effivity food safety management software provides a comprehensive set of tools, modules and capabilities to help manage food safety efficiently and effectively.
Our workflow-driven ISO 22000 FSMS software ensures compliance management and corporate governance in your organisation by unifying:
✓ Communication
✓ Compliance obligations
✓ Processes and procedures
✓ Risks and opportunities
✓ PRPs
✓ Hazard control
✓ Logistics
✓ Customer experience
Benchmark your FSMS with the best global food safety practices, pass the most stringent food safety standards in the world and ensure both productivity and quality are consistently high. inspections and audits of various certification bodies.
Our software covers all modules required to implement, certify and maintain an effective FSMS and combine quality with compliance.
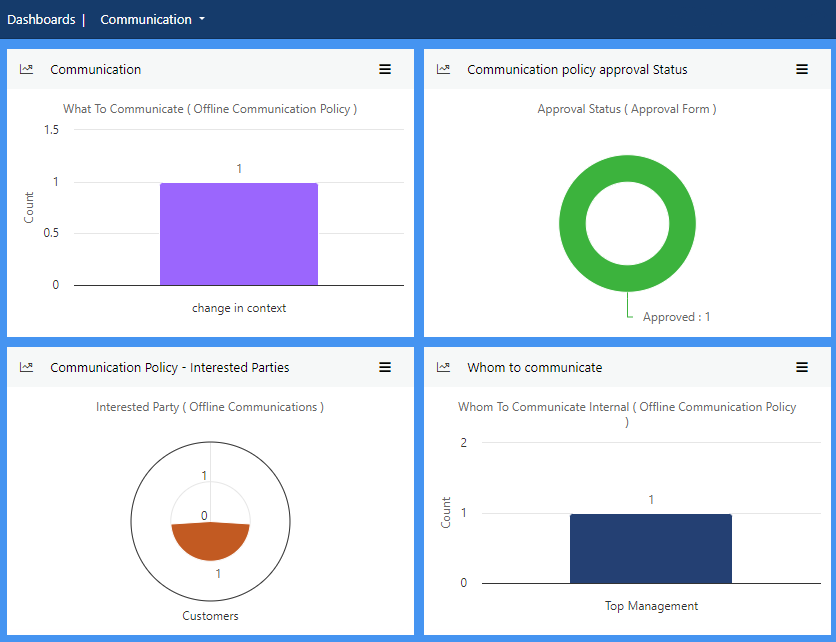
Communication is essential for all components of your FSMS. Automate tasks and follow-up, collaborate across departments and ensure food safety remains at the heart of what you do through clear, unambiguous, timely communication.
This is particularly beneficial for multi-site organisations.
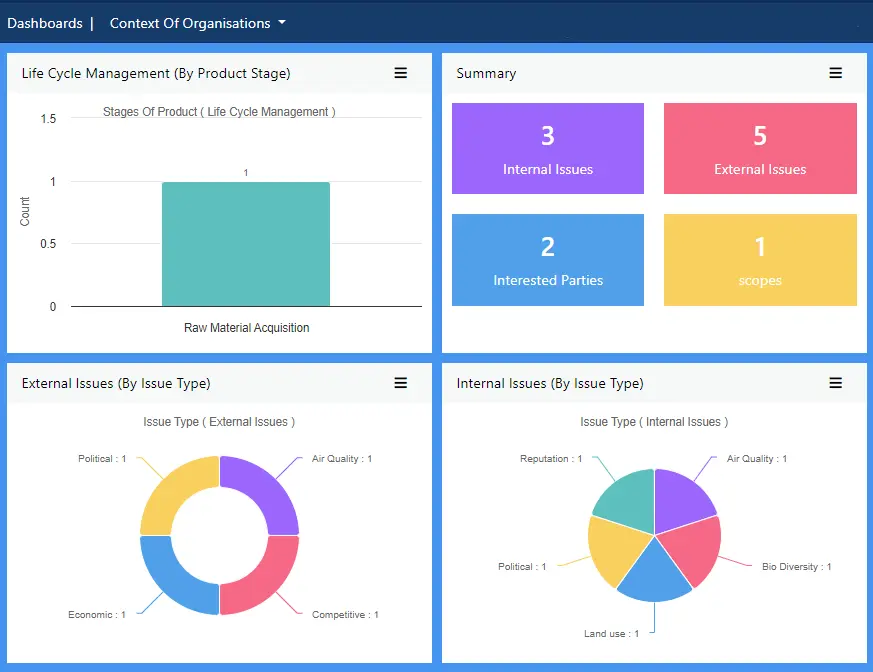
View more...Align food safety management with the strategic aims of your business, ensuring the two are compatible with one another and not having to compete for supremacy. Define and manage internal and external issues from the perspective of food safety.
View more...This module provides a comprehensive and collaborative way to manage food safety management across your organisation.
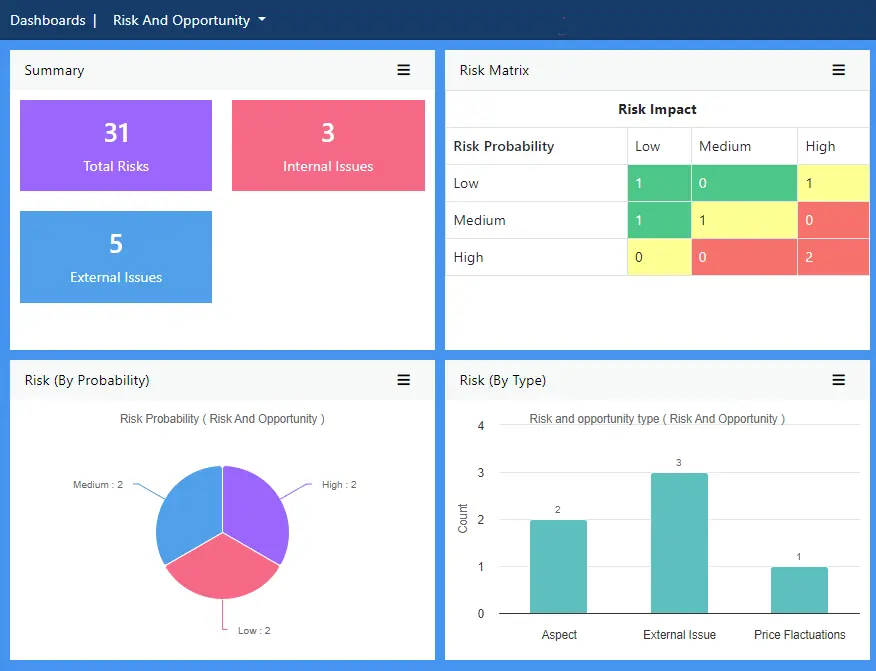
Identify, review, mitigate and control FSMS risks to future-proof your business with all. FSMS threats can be documented and held alongside other business risk data for a comprehensive repository that ensures a consistent approach to protecting the organisation.
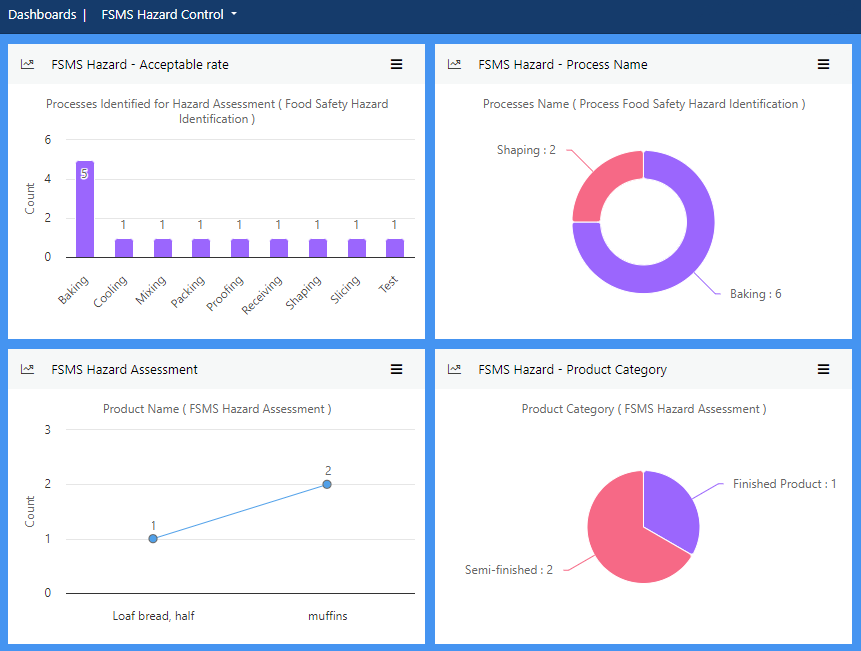
View more...Manage all aspects of hazard control to ensure you consistently meet statutory, regulatory and customer requirements. Conduct hazard assessments to determine the likelihood, possible severity and likely frequency of potential hazards.
Use this module to plan, implement and document your Operational Prerequisite Program (OPRP) or as Critical Control Point (CCP).
Define the characteristics of raw materials, ingredients, contact materials and finished products. Create a process flow diagram and identify potential hazards associated with each step.
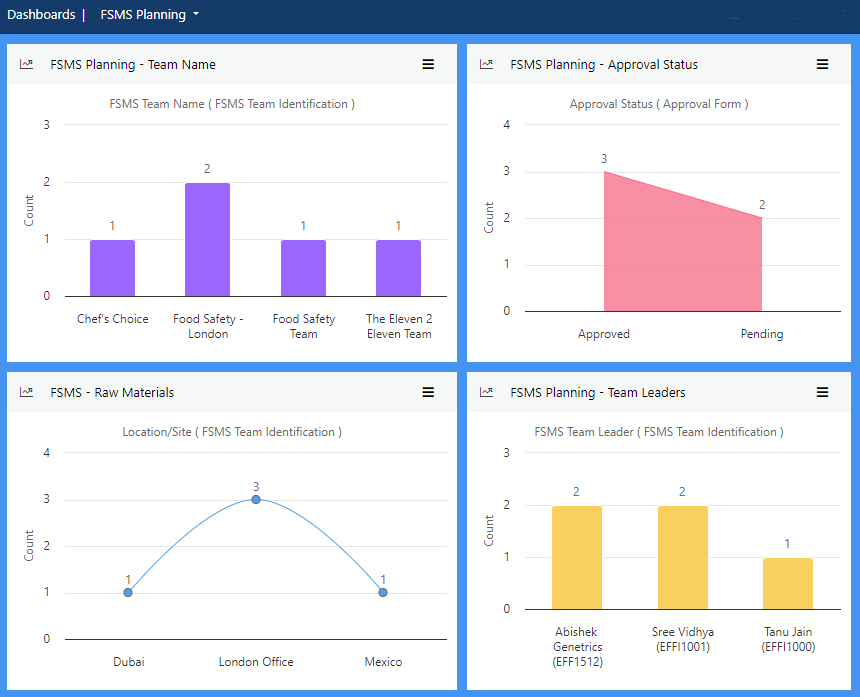
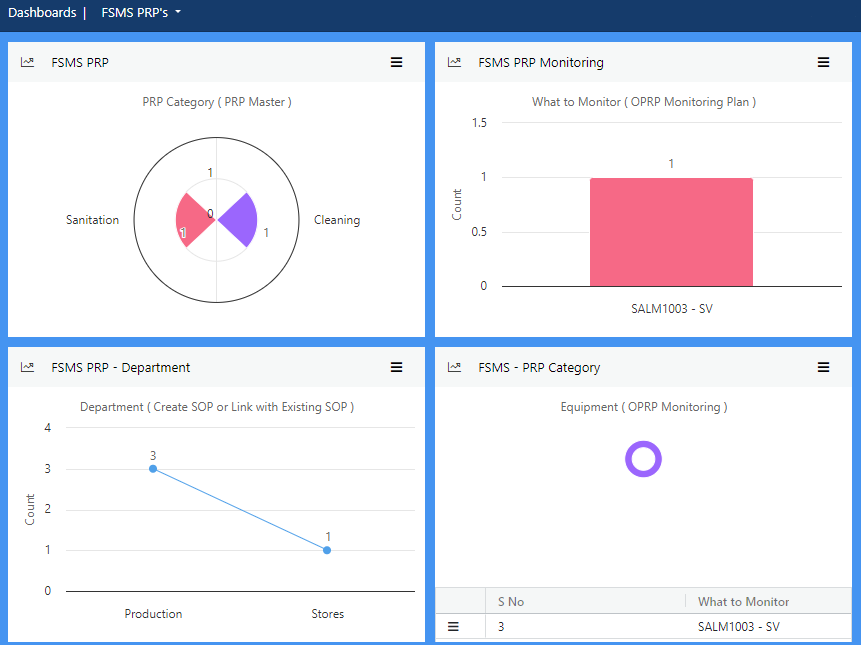
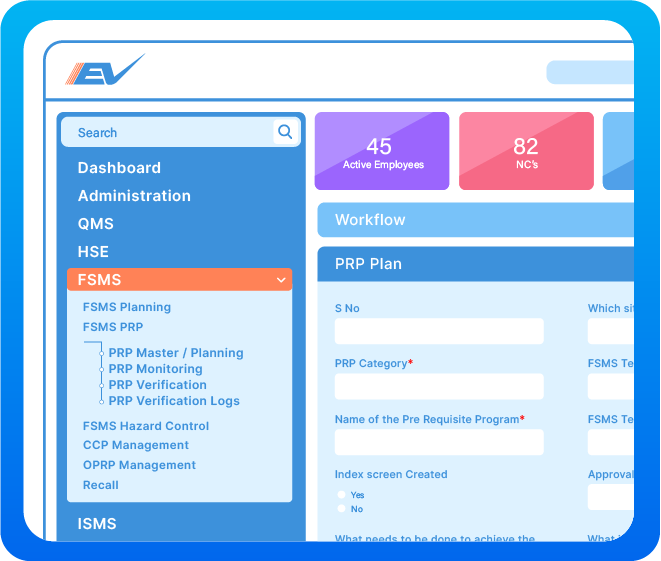
Establish Prerequisite Programs (PRPs) to minimise the risk of food safety hazards in your products, and processes. Plan monitoring and verification at regular intervals to ensure consistency.
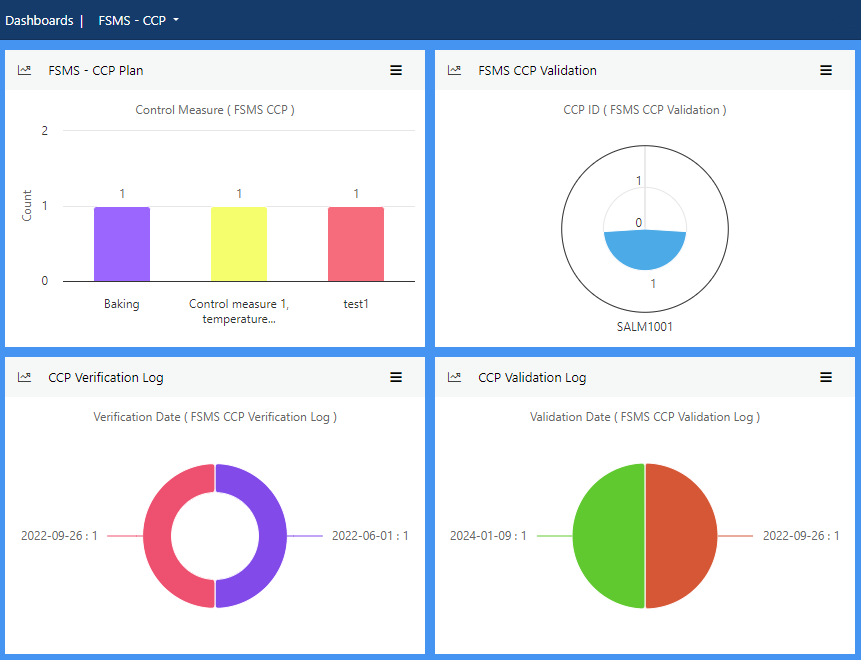
Manage documentation of Critical Control Point (CCP) through defining critical limits, monitoring activities and CCP deviation plan as well as planning for verification and validation activities. Capture data to record results of CCP monitoring, verification and validation activities.
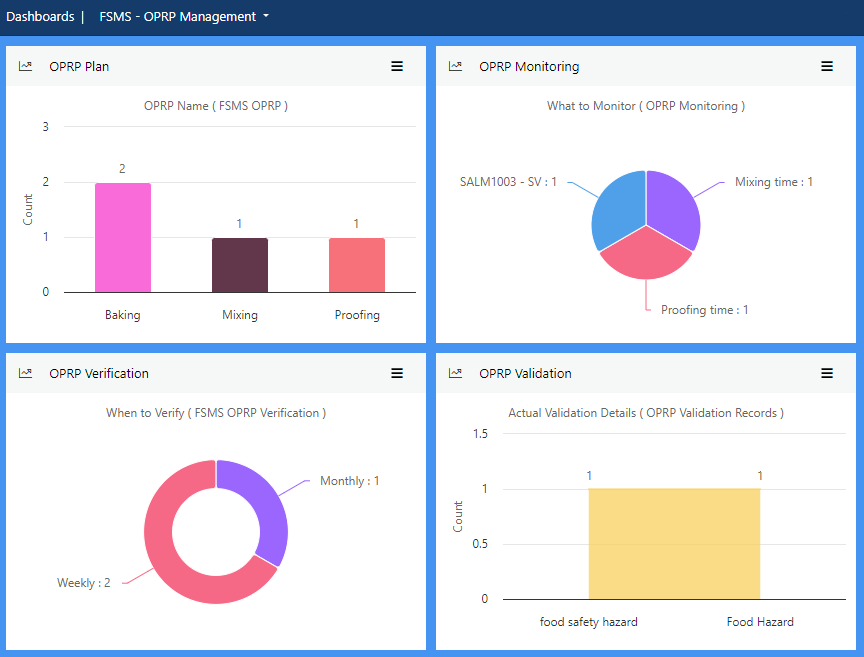
Manage documentation of Operational Prerequisite Program (OPRP) by defining criteria, monitoring activities and OPRP deviation plan, and developing verification and validation activities. Capture real time data to record results of OPRP monitoring, verification and validation activities.
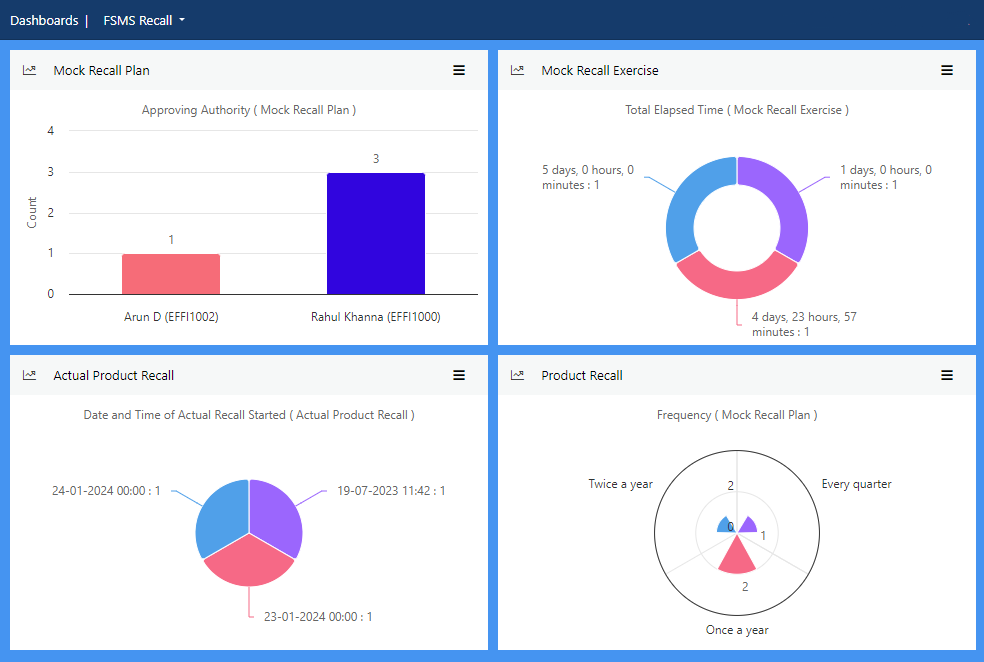
Manage preparedness for instances of product withdrawal or recall that could impact your organisation’s role in the food chain.
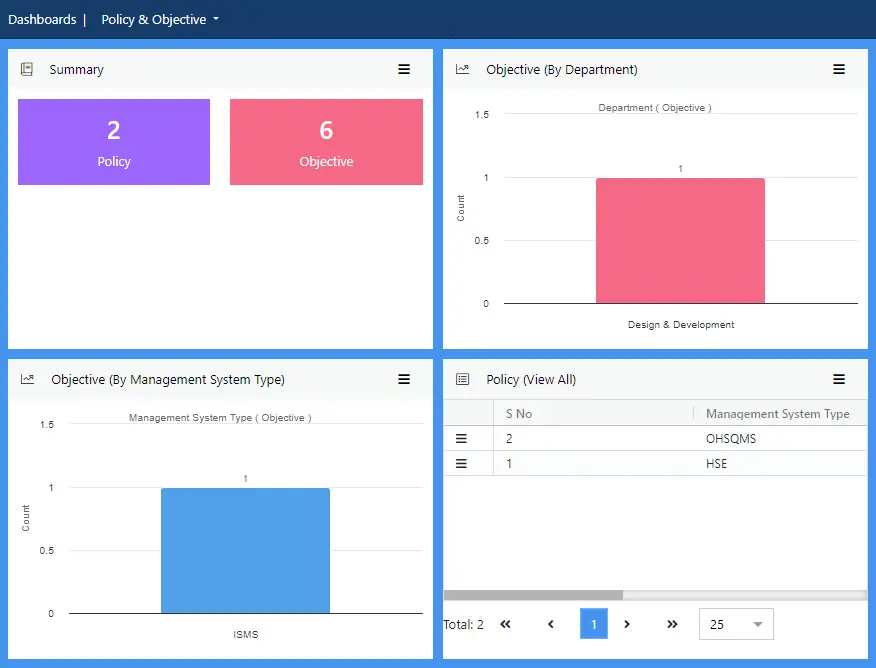
Create synergy between your vision, mission and purpose, and the intended outcome of your FSMS. Create, discuss, approve and publish policies quickly and efficiently. Once the objectives of the FSMS are approved, a detailed plan can be created, and performance tracked regularly.
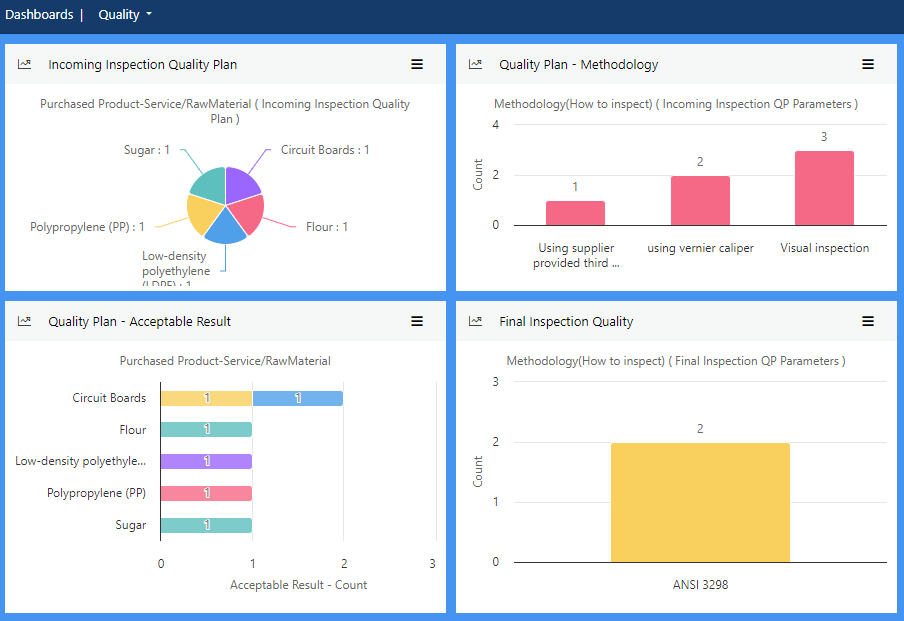
View more...Make inspection, internal audits and quality control user-friendly with this module that automates inspections and collates real-time data concerning non-conformities.
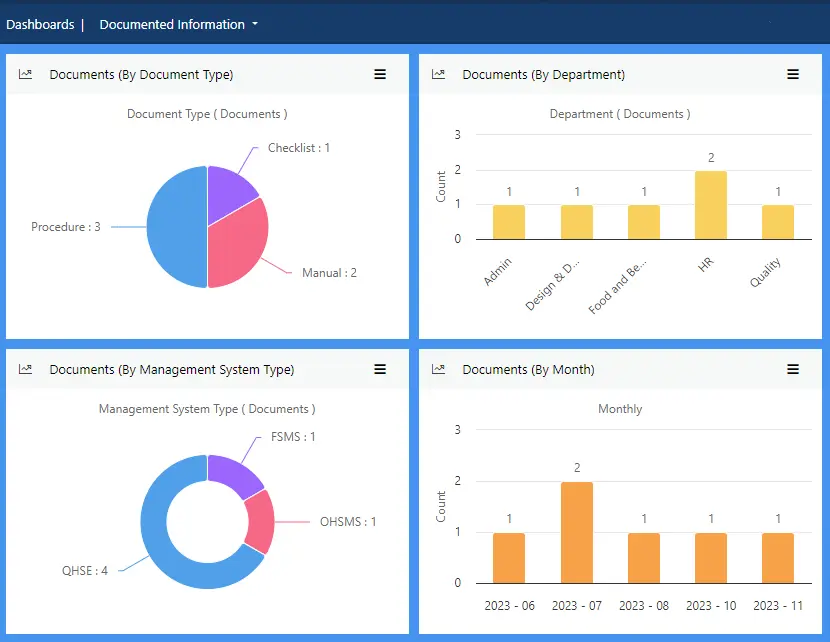
View more...Store and manage all your food safety documentation in one place to ensure you meet ISO 22000:2018 standards.
A central repository of documents ensures all authorised users have access to relevant documents from any location at any time. A robust change management process allows users to initiate change requests for review, approval, rejection or revision with comments. Upon approval, revision numbers are assigned in auto increments and old documents are archived automatically.
Effivity enables the management of internally and externally produced documents.
View more...Manage hazard control deviation, withdrawal/recall, process deviations, product and service-related problems, customer complaints, deviations from compliance obligations, customer requirements and ISO standards and more with this module.
If not managed effectively these types of issues can have a long-term negative impact on reputation, profitability and business continuity, so a robust, user-friendly platform for managing them is essential.
Storing all non-conformity data and information in one place enables you to obtain comprehensive insights and take positive action to prevent recurrence.
New non-conformities can be created and assigned to users in this module. Capture corrections, root cause analysis, corrective actions, and monitor effectiveness evaluation of interventions.
Define the criticality of the non-conformity and any costs associated with resolution. The access control feature ensures only authorised personnel have access to non-conformities.
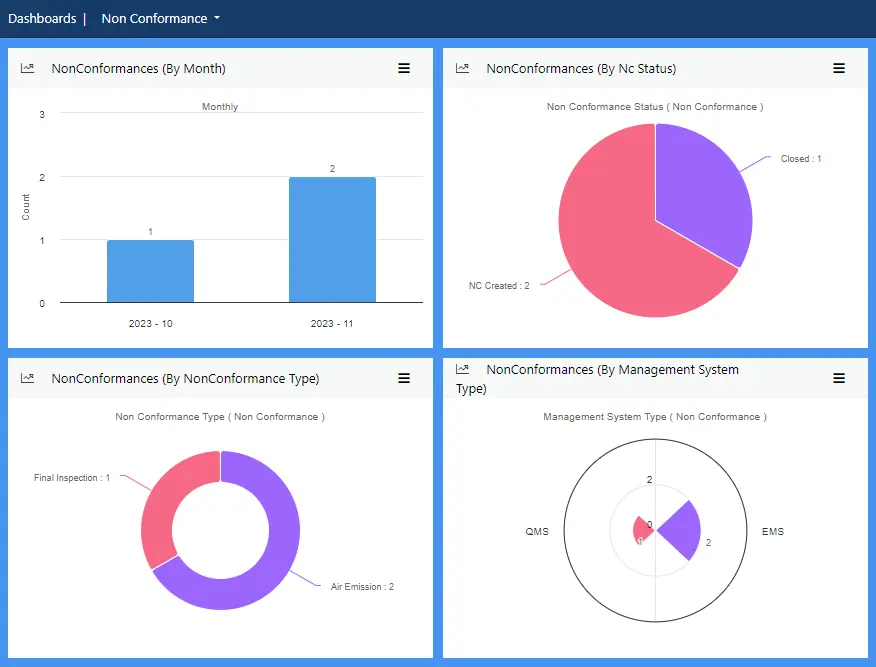
Dashboards provide real-time data analysis and statistical evaluation of non-conformities across various parameters.
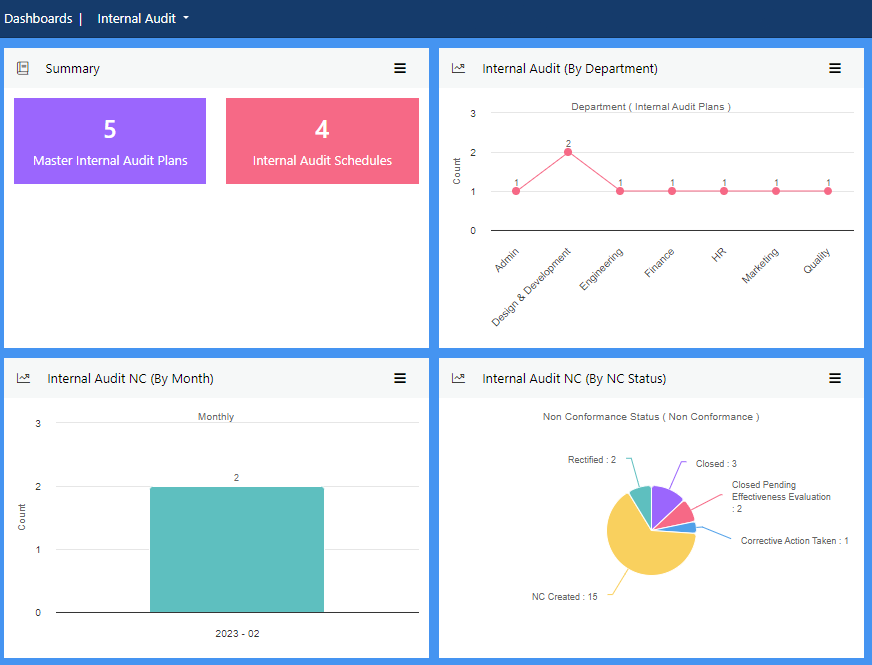
View more...The most effective way to achieve and maintain compliance is to deploy a stringent internal audit policy that replicates industry standards.
Create a master internal audit plan and drill it down to a micro level as required. Devise bespoke internal audit checklists or use pre-configured checklists provided free with your Effivity subscription.
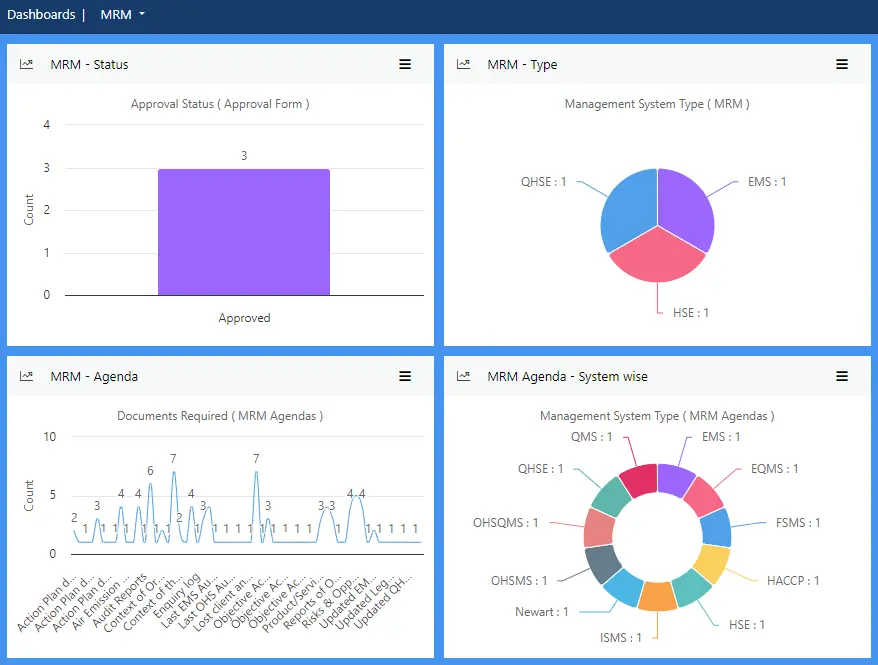
View more...Management Review allows business leaders to instantly get a snapshot of food safety standards to check for suitability, effectiveness and alignment with the strategic direction of the organisation.
Schedule management reviews, create an agenda, invite participants, record minutes and create an action plan through this module. Then track progress against the action plan quickly and easily from any device.
View more...